Unlocking the Invisible
Inside the UW School of Pharmacy’s Mass Spectrometry Center
By Scott Braswell
UW School of Pharmacy
When Dale Whittington arrived at the University of Washington, he did not imagine that he would one day help shape one of the School of Pharmacy’s most consequential research facilities. Trained as a biologist, with a career path that briefly veered away from science altogether, Whittington eventually found his way back – drawn by the chance to work at the intersection of discovery, teaching, and problem-solving.
More than a decade later, as technical director and manager of the UW School of Pharmacy’s Mass Spectrometry Center, he oversees an operation that quietly underpins a wide range of scientific work. It is not a space defined by spectacle, but by precision: instruments that weigh molecules with extraordinary accuracy, revealing details that would otherwise remain invisible.
“This is the tool that lets us interrogate biological and chemical questions at a fundamental level, whether you’re studying drug concentrations in tissue or changes in proteins during disease, mass spectrometry gives you a way to see what’s actually happening.”

Dale Whittington
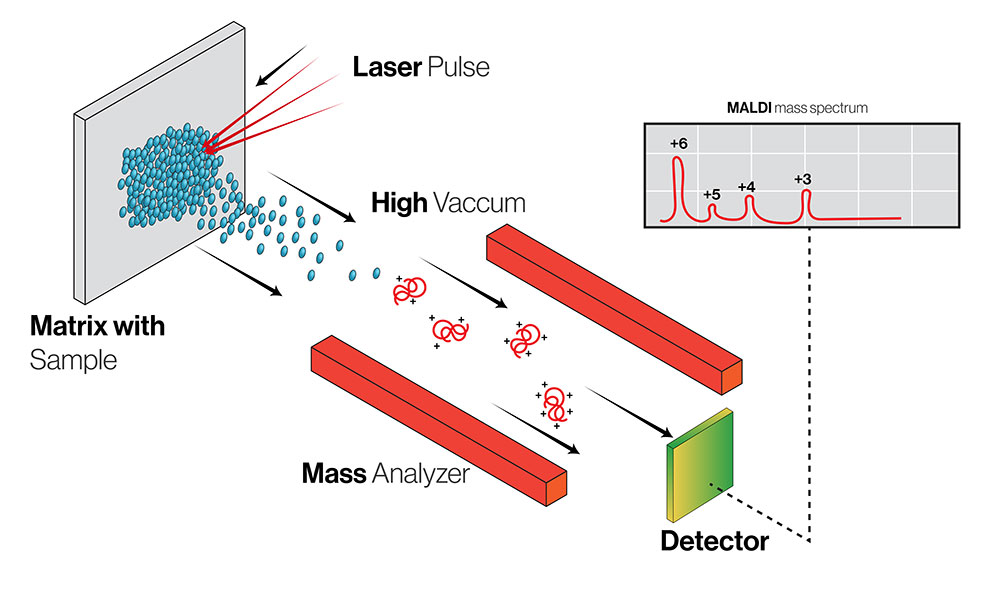
At its core, mass spectrometry rests on a deceptively simple principle. Molecules are given an electrical charge and guided through electromagnetic fields toward a detector. How they move—and how long they take to arrive—reveals their identity, structure, and abundance. From this information, researchers can determine molecular weights, concentrations, and subtle chemical differences that shape how drugs behave in the body.
From small molecules like amino acids to complex proteins and antibodies, mass spectrometry has become a foundational tool across chemistry, biology, and medicine. Its reach is broad, but its value lies in specificity.
From Molecules to Meaning
The real power of mass spectrometry emerges not from the instrument itself, but from the questions it allows scientists to ask. Researchers use it to track how medications are absorbed, distributed, and eliminated; to study how proteins change in disease; and to understand why a therapy may work well in one context but fail in another.

“This is the tool that lets us interrogate biological and chemical questions at a fundamental level,” Whittington says. “Whether you’re studying drug concentrations in tissue or changes in proteins during disease, mass spectrometry gives you a way to see what’s actually happening.”
For Mary Hebert, a professor of pharmacy and director of the UW’s Obstetric-Fetal Pharmacology Research Unit, the Center’s work is essential to understanding how medications behave during pregnancy and lactation—populations historically underrepresented in clinical research.
“The pharmacokinetics of most medications have not been studied in these populations,” she explains. “The Mass Spectrometry Center has made it possible to identify not only how drug handling changes during pregnancy, but also allows us to make informed predictions about other medications eliminated via similar pathways.”
Learning by Doing
What distinguishes the UW School of Pharmacy’s Mass Spectrometry Center is not only its technical capacity, but its approach to training. At many institutions, mass spectrometers are operated exclusively by specialists, with users receiving only processed data. At UW, students are encouraged to engage directly with the instruments themselves.
“Our goal is to train mass spectrometrists, not just data recipients,” Whittington says. “Students learn how to prepare samples, optimize methods, troubleshoot problems, and design experiments from start to finish.”

Dale Whittington and Medicinal Chemistry graduate student, Corrina Cooper.
“Direct access to mass spectrometry empowers [graduate students] to generate and interpret complex datasets independently. That accelerates their development as scientists and makes them more competitive for fellowships and careers.”
Graduate students progress quickly from observation to hands-on work. By their third session, many are expected to operate independently, with staff stepping in only when needed. The experience demystifies the technology—and builds confidence.
That experience carries weight beyond the university. Graduates trained at the Center enter industry and academic labs able to operate instruments, interpret results, and adapt methods with minimal supervision. Employers recognize the difference.
Medicinal Chemistry Affiliate Assistant Professor Kim Alonge, whose research focuses on the brain’s extracellular matrix, sees that impact firsthand in her trainees. “Direct access to mass spectrometry empowers them to generate and interpret complex datasets independently,” she says. “That accelerates their development as scientists and makes them more competitive for fellowships and careers.”
Expanding the Boundaries
While pharmaceutical research remains central to the Center’s mission, its applications extend far beyond drug development. Mass spectrometry has been used to analyze compounds in cannabis products, verify the composition of fish oil supplements, and even fingerprint wines to confirm their geographic origins.

One rapidly advancing area is MALDI imaging, a technique that allows researchers to map the spatial distribution of molecules within thin slices of tissue. The sample remains intact, preserving information about where compounds accumulate—or fail to reach.
This approach has reshaped understanding of HIV therapies by revealing uneven drug penetration in tissues, helping explain how viral reservoirs persist despite treatment. In neuroscience, it is opening new windows into the chemistry of the brain.

Using the Bruker tims TOF fleX platform, the only open access MALDI (matrix assisted laser desorption ionization) tissue imaging mass spectrometry in the northwest, UW students and researchers working with Alonge have mapped glycans in the hippocampus, uncovering spatial patterns linked to Alzheimer’s disease. “Without access to this facility—and this specific technology—those insights would not have been possible,” she says.
Looking ahead, Whittington sees artificial intelligence playing a growing role in managing the sheer volume of data mass spectrometry generates. “There’s more information than any one person can reasonably process,” he notes. “If we can apply AI in a careful, unbiased way, it may reveal patterns we’re not yet able to see.”
“There are critical questions—especially in pregnancy—that simply can’t be answered without highly sensitive, specific assays. Without this Center, we would not be able to quantify drug exposures that matter for both mother and child.”
A Place to Grow
For Whittington, the appeal of the Center lies in its constant motion. Each project presents a new puzzle; each student brings a different way of thinking. The work resists routine.
“I feel fortunate to do something that’s always changing,” he says. “Between the teaching, the experiments, and the problem-solving, no two days are the same.”
For students and researchers alike, the Mass Spectrometry Center is more than a shared resource. It is a place where theory meets practice, where complex tools become accessible, and where careful measurement leads to clearer understanding.
As Hebert puts it, “There are critical questions—especially in pregnancy—that simply can’t be answered without highly sensitive, specific assays. Without this Center, we would not be able to quantify drug exposures that matter for both mother and child.”
In a field often defined by what cannot yet be seen, the Mass Spectrometry Center helps bring the invisible into focus—one molecule at a time.
Scott Braswell is the Director of Marketing and Communications in the UW School of Pharmacy.
This article was originally published at the School of Pharmacy website. Republished with permission.